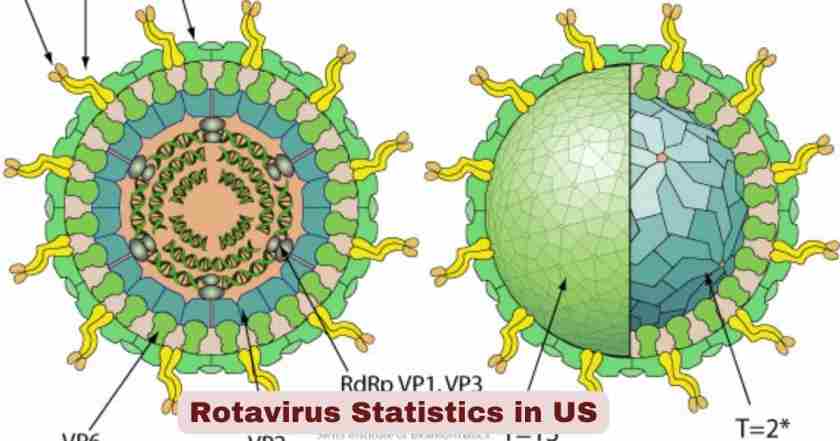
What is Rotavirus?
Rotavirus remains one of the most significant vaccine-preventable diseases affecting American infants and young children, despite substantial progress made since the introduction of rotavirus vaccines in 2006. The rotavirus statistics in the US 2026 demonstrate both remarkable achievements in disease prevention and ongoing challenges in achieving optimal vaccination coverage. Understanding rotavirus in America 2026 requires examining comprehensive data on infection rates, vaccination coverage, symptoms, and the dramatic public health impact of immunization programs across the United States.
Rotavirus gastroenteritis continues to affect thousands of American children annually, though at dramatically reduced rates compared to the pre-vaccine era when nearly every child experienced rotavirus infection before their fifth birthday. The latest CDC rotavirus statistics 2026 reveal current trends in disease burden, vaccine effectiveness, and the critical importance of maintaining high vaccination rates. This comprehensive analysis of rotavirus disease statistics in the US 2026 provides essential insights for parents, healthcare providers, and public health officials working to protect vulnerable infant populations from this highly contagious viral infection.
Interesting Facts About Rotavirus in the US 2026
| Key Rotavirus Facts | 2026 Statistics |
|---|---|
| Pre-Vaccine Annual Hospitalizations | 55,000-70,000 children |
| Pre-Vaccine Annual ED Visits | 205,000-272,000 visits |
| Pre-Vaccine Annual Doctor Visits | 410,000 visits |
| Post-Vaccine Hospitalization Reduction | 80% decrease |
| Post-Vaccine ED Visit Reduction | 57% decrease |
| Current Vaccine Coverage Rate | 71.8% complete series |
| Vaccine Effectiveness (Hospitalizations) | 84-89% |
| Vaccine Effectiveness (ED Visits) | 79-83% |
| Incubation Period | 1-3 days |
| Symptom Duration | 3-8 days |
| Infectious Dose | <100 virus particles |
| Annual Hospitalizations Prevented | 40,000-50,000 cases |
Data Source: Centers for Disease Control and Prevention, National Center for Immunization and Respiratory Diseases, 2025-2026
The rotavirus statistics in America 2026 reveal extraordinary public health achievements while highlighting areas requiring continued attention. The 80% reduction in hospitalizations since vaccine introduction represents approximately 40,000 to 50,000 prevented hospitalizations annually, saving thousands of children from severe dehydration and potentially life-threatening complications. The vaccine effectiveness of 84-89% against hospitalizations demonstrates robust protection, though the current coverage rate of 71.8% falls short of the Healthy People 2030 goal of 80% and lags 15-20 percentage points behind other routine childhood vaccinations.
The highly contagious nature of rotavirus is evident in its infectious dose of fewer than 100 virus particles, making it extremely efficient at spreading through daycare centers, households, and communities. The 1-3 day incubation period followed by 3-8 days of symptoms creates significant challenges for containment without vaccination. Recent 2026 data shows vaccination rates declined by 1.7 percentage points for rotavirus compared to previous years, emphasizing the need for renewed public health efforts to maintain and improve immunization coverage across all demographic groups.
Rotavirus Vaccination Coverage in the US 2026
| Vaccination Coverage Category | 2026 Rate | Comparison to Other Vaccines |
|---|---|---|
| Complete Rotavirus Series | 71.8% | 15-20% lower than DTaP |
| DTaP Vaccine Coverage (≥3 doses) | 94-95% | Significantly higher |
| Polio Vaccine Coverage (≥3 doses) | 93% | Significantly higher |
| MMR Vaccine Coverage (≥1 dose) | 91% | Significantly higher |
| Pneumococcal Coverage (PCV) | 90-92% | Significantly higher |
| Rotavirus Coverage Decline (2025-2026) | -1.7 percentage points | Recent decrease |
| Healthy People 2030 Goal | 80% target | Not yet achieved |
| American Indian/Alaska Native Coverage | 61.5% | Lowest demographic |
Data Source: CDC National Immunization Survey-Child, March 2026 Report
Rotavirus vaccination coverage in America 2026 demonstrates concerning disparities, with the 71.8% complete series rate representing a 1.7 percentage point decline from previous years. This decrease occurred alongside declines in other childhood vaccines including influenza (7.4 percentage point drop), hepatitis B birth dose (1.8 percentage points), and pneumococcal conjugate vaccine (1.5 percentage points). The persistent gap between rotavirus vaccination and other routine childhood immunizations like DTaP at 94-95% coverage reflects unique challenges in rotavirus vaccine administration.
Age restrictions for rotavirus vaccination create significant barriers to achieving higher coverage rates. The maximum age for the first dose is 14 weeks and 6 days, with the final dose required before 8 months of age, leaving healthcare providers with limited windows for catch-up vaccination compared to other childhood vaccines. Racial and ethnic disparities are particularly evident among American Indian and Alaska Native children with only 61.5% receiving rotavirus vaccination, compared to higher rates in other demographic groups. Geographic variations between metropolitan and non-metropolitan areas show 1-2 percentage point lower coverage in rural communities, compounding healthcare access challenges in underserved regions.
Pre-Vaccine Rotavirus Burden in the US 2026
| Pre-Vaccine Disease Burden | Annual Statistics | Impact |
|---|---|---|
| Total Infections by Age 5 | Nearly 100% of children | Universal exposure |
| Hospitalizations Annually | 55,000-70,000 cases | Severe dehydration |
| Emergency Department Visits | 205,000-272,000 visits | Acute care burden |
| Physician Office Visits | 410,000 visits | Primary care strain |
| Annual Deaths | 20-60 deaths | Preventable mortality |
| Annual Economic Cost | $1 billion | Healthcare expenses |
| Median Hospitalization Cost | $3,581 per child | Financial burden |
| Median ED Visit Cost | $574 per child | Out-of-pocket expenses |
Data Source: CDC Historical Surveillance Data, Pre-2006 Era
Before rotavirus vaccine introduction in 2006, rotavirus was the leading cause of severe gastroenteritis in American infants and young children, with nearly every child infected by their fifth birthday. The annual burden of 55,000-70,000 hospitalizations represented enormous strain on pediatric healthcare systems, particularly during winter and spring months when rotavirus activity peaked. Emergency departments managed 205,000-272,000 visits annually, with children presenting with severe dehydration requiring intravenous fluid therapy and intensive monitoring.
Primary care physicians conducted 410,000 office visits for rotavirus gastroenteritis, though many mild cases never reached medical attention. The economic impact exceeded $1 billion annually when accounting for direct medical costs, parental work absences, and indirect societal expenses. Fatal outcomes occurred in 20-60 children annually, primarily among those with underlying medical conditions or those who developed severe complications before receiving appropriate medical intervention. The universal nature of rotavirus exposure in the pre-vaccine era meant that improved hygiene and sanitation alone proved insufficient to reduce disease burden significantly.
Post-Vaccine Rotavirus Impact in the US 2026
| Post-Vaccine Impact Category | Reduction Achieved | Current Status |
|---|---|---|
| Hospitalization Reduction | 80% decrease | Median across studies |
| Emergency Department Reduction | 57% decrease | Sustained decline |
| Annual Hospitalizations Prevented | 40,000-50,000 cases | Ongoing benefit |
| Healthcare Cost Reduction | $121-231 million annually | Economic savings |
| Overall Disease Burden | Dramatically reduced | Population-level impact |
| Biennial Pattern Emergence | Odd/even year cycling | Epidemiologic change |
| Indirect Protection (Unvaccinated) | Significant herd immunity | Community benefit |
| Strain Distribution Changes | G12P[8] predominance | Vaccine coverage maintained |
Data Source: CDC National Respiratory and Enteric Virus Surveillance System, 2006-2026
Rotavirus vaccine introduction resulted in a median 80% decline in rotavirus-associated hospitalizations and a 57% decrease in emergency department visits, representing one of the most successful vaccine programs in recent American public health history. The 40,000-50,000 hospitalizations prevented annually translate to thousands of children spared from severe dehydration, electrolyte imbalances, and potential complications. Economic analyses demonstrate annual healthcare cost reductions of $121-231 million, benefiting families, insurance systems, and public health programs.
A unique biennial pattern emerged post-vaccination, with higher rotavirus incidence during odd-numbered years and lower incidence during even-numbered years. This pattern reflects annual fluctuations in susceptible children and transmission efficiency, predicted by epidemiologic models studying vaccination coverage below 80%. Indirect protective benefits extend beyond vaccinated children, with unvaccinated age groups and household members experiencing reduced rotavirus exposure due to decreased community transmission. Strain surveillance shows G12P[8] has become predominant among severe cases, though vaccine effectiveness remains high across all circulating genotypes.
Rotavirus Vaccine Types in the US 2026
| Vaccine Characteristic | RotaTeq (RV5) | Rotarix (RV1) |
|---|---|---|
| Manufacturer | Merck & Company | GlaxoSmithKline |
| Vaccine Type | Pentavalent (5 strains) | Monovalent (1 strain) |
| Strains Covered | G1, G2, G3, G4, P[8] | G1P[8] |
| Dosing Schedule | 3 doses | 2 doses |
| Recommended Ages | 2, 4, 6 months | 2, 4 months |
| First Dose Maximum Age | 14 weeks 6 days | 14 weeks 6 days |
| Final Dose Maximum Age | 8 months 0 days | 8 months 0 days |
| Efficacy (Severe Disease) | 98% | 96% |
| Efficacy (Any Severity) | 74% | 87% |
| Hospitalization Prevention | 96% | 96% |
| ED Visit Prevention | 94% | High efficacy |
Data Source: CDC Advisory Committee on Immunization Practices, Vaccine Recommendations
Two rotavirus vaccines are licensed and recommended in the United States: RotaTeq (RV5), a pentavalent vaccine containing five reassortant strains, and Rotarix (RV1), a monovalent vaccine containing one attenuated strain. RotaTeq requires three doses administered at 2, 4, and 6 months of age, while Rotarix uses a two-dose schedule at 2 and 4 months. Both vaccines demonstrate exceptional efficacy, with RotaTeq showing 98% effectiveness and Rotarix showing 96% effectiveness against severe rotavirus gastroenteritis.
Clinical trials involving over 70,000 infants for RotaTeq and 63,000 infants for Rotarix established robust safety and efficacy profiles. Both vaccines reduced hospitalization risk by 96% and emergency department visits by 94%, with protection extending across multiple rotavirus genotypes beyond those specifically targeted in vaccine formulations. ACIP recommendations allow interchangeability between vaccines when the initial product is unavailable, though completion with the same vaccine is preferred. The strict age restrictions requiring first doses before 14 weeks 6 days and final doses before 8 months aim to minimize intussusception risk observed in post-licensure surveillance.
Rotavirus Symptoms in the US 2026
| Symptom Category | Characteristics | Duration/Severity |
|---|---|---|
| Primary Symptom | Watery diarrhea | 3-8 days typical |
| Gastrointestinal | Vomiting (often first symptom) | Precedes diarrhea |
| Fever | Temperature >102°F (>39°C) | Up to 33% of patients |
| Dehydration | Severe fluid loss | Life-threatening risk |
| Abdominal Pain | Cramping and discomfort | Common symptom |
| Loss of Appetite | Reduced oral intake | Worsens dehydration |
| Incubation Period | Asymptomatic phase | 1-3 days |
| Illness Onset | Abrupt symptom start | Sudden onset |
| Recovery Timeline | Symptom resolution | 3-7 days typically |
Data Source: CDC Clinical Overview of Rotavirus, National Vital Statistics
Rotavirus illness ranges from mild watery diarrhea to severe diarrhea with vomiting and fever that can result in dehydration, shock, electrolyte imbalance, and on rare occasions for American children, death. The classic presentation begins abruptly after a 1-3 day incubation period, with vomiting often preceding diarrhea onset. The watery diarrhea can be profuse, leading to rapid fluid loss particularly dangerous in infants and young children with limited fluid reserves.
Fever occurs in approximately one-third of patients, with temperatures exceeding 102°F (39°C) adding to fluid loss through increased metabolic demands. Dehydration represents the most serious complication, manifesting as decreased urination, dry mucous membranes, sunken eyes, lethargy, and in severe cases altered consciousness. Abdominal pain and cramping accompany the gastrointestinal symptoms, while loss of appetite further complicates efforts to maintain adequate hydration through oral intake. Symptom duration typically spans 3-7 days, though complete recovery may require additional time for bowel function normalization and nutritional rehabilitation in severely affected children.
Rotavirus Transmission in the US 2026
| Transmission Route | Mechanism | Risk Factors |
|---|---|---|
| Fecal-Oral Route | Primary transmission | Contaminated hands |
| Person-to-Person Contact | Direct contact | Close contact settings |
| Fomite Transmission | Contaminated surfaces | Daycare environments |
| Respiratory Droplets | Possible route | Airborne particles |
| Contaminated Food/Water | Foodborne transmission | Hygiene failures |
| High Viral Shedding | Stool concentration | Infectious period |
| Environmental Stability | Virus persistence | Survives on surfaces |
| Daycare Centers | Amplification sites | Multiple exposures |
| Household Spread | Family clusters | Sibling transmission |
Data Source: CDC Rotavirus Transmission Epidemiology
Rotaviruses are shed in high concentrations in stools of infected persons and transmitted primarily by the fecal-oral route, both through close person-to-person contact and through fomites (contaminated objects and surfaces). The extremely low infectious dose of fewer than 100 virus particles makes rotavirus exceptionally contagious, with transmission occurring readily in daycare centers, households, and healthcare facilities. Viral shedding begins before symptom onset and continues throughout the illness period, enabling asymptomatic and mildly symptomatic individuals to spread infection.
Fomite transmission occurs when children touch contaminated toys, doorknobs, changing tables, and other surfaces before putting hands in mouths. Environmental stability allows rotavirus to persist on surfaces for extended periods, defeating routine cleaning efforts without appropriate disinfection protocols. Respiratory droplet and fecally contaminated food and water represent additional transmission routes, though less common than direct fecal-oral spread. Daycare centers serve as major amplification sites, with multiple simultaneous exposures creating ideal conditions for transmission. Household spread rapidly infects siblings and occasionally parents, though adult infections typically produce milder symptoms due to previous exposures building partial immunity.
Rotavirus Age Distribution in the US 2026
| Age Group | Infection Risk | Severity Profile |
|---|---|---|
| 3-35 Months | Highest risk period | Most severe disease |
| Infants <12 Months | Very high risk | Severe dehydration |
| Toddlers 12-35 Months | High risk | Hospitalization risk |
| Children 3-5 Years | Moderate risk | Generally milder |
| School-Age Children | Lower risk | Mild symptoms |
| Adults | Occasional infection | Usually asymptomatic/mild |
| Elderly Adults | Vulnerable population | Can be severe |
| Immunocompromised | All ages at risk | Potentially fatal |
| First Infection | Most severe | Primary immunity |
Data Source: CDC Age-Stratified Rotavirus Surveillance Data
Severe dehydrating rotavirus infection occurs primarily among children 3-35 months of age, representing the peak vulnerability period for life-threatening complications. Infants younger than 12 months face particular risks due to small body size, limited fluid reserves, and difficulty communicating thirst or early dehydration symptoms. Toddlers aged 12-35 months continue experiencing high rates of severe disease, though developing partial immunity from initial exposures provides some protection against subsequent severe episodes.
Natural history studies demonstrate that repeated rotavirus exposures throughout the human lifespan create progressive reduction in symptomatic illness risk with each subsequent infection, explaining why older children and adults typically experience milder disease. First infections produce the most severe symptoms, while second and third infections generally cause progressively milder illness. Immunocompromised children can experience severe, prolonged, and even fatal rotavirus gastroenteritis regardless of age. Adult infections usually remain asymptomatic or mild, though elderly adults and those with underlying medical conditions may develop severe gastroenteritis requiring medical intervention.
Rotavirus Vaccine Effectiveness in the US 2026
| Effectiveness Measure | RotaTeq (RV5) | Rotarix (RV1) |
|---|---|---|
| Full Series – Hospitalizations | 85% (95% CI: 80-87%) | 83% (95% CI: 72-89%) |
| Full Series – ED Visits | 79% (95% CI: 75-83%) | Similar effectiveness |
| Any Dose (≥1) – Hospitalization/ED | 78% (95% CI: 75-80%) | Combined estimate |
| Against Severe Disease | 94% (95% CI: 90-97%) | Very high protection |
| Against Mild Disease | 59% (95% CI: 49-67%) | Moderate protection |
| Children <3 Years | Highest effectiveness | Peak protection |
| Older Children | Decreasing with age | Waning immunity |
| G1-G4 Strains | 74-98% effectiveness | Excellent coverage |
| Cross-Protection | Effective against G12P[8] | Broad protection |
Data Source: CDC Meta-Analysis 2006-2022, NVSN Surveillance Data
Vaccine effectiveness studies spanning 2006-2017 found full-series effectiveness for RotaTeq at 85% (95% CI: 80-87%) and Rotarix at 83% (95% CI: 72-89%) against rotavirus-associated hospitalizations and emergency department visits. More recent 2009-2022 analyses showed any dose (≥1) effectiveness of 78% (95% CI: 75-80%) against rotavirus-associated ED visits or hospitalizations, with protection ranging from 59% for mild disease to 94% for very severe disease requiring intensive medical intervention.
Age-stratified analyses demonstrate highest vaccine effectiveness among children under three years, with protection decreasing as children age beyond this peak vulnerability period. Effectiveness against specific genotypes shows 74% protection against any severity of G1-G4 rotavirus gastroenteritis and 98% against severe disease in targeted strains. Cross-protection extends to non-vaccine strains including G12P[8], which became predominant in recent years, demonstrating the broad immunologic response generated by current vaccines. Real-world effectiveness consistently matches or exceeds clinical trial efficacy, confirming robust protection under diverse field conditions.
Rotavirus Seasonal Patterns in the US 2026
| Seasonal Characteristic | Pattern Description | Geographic Variation |
|---|---|---|
| Pre-Vaccine Seasonality | Winter-spring peaks | December-May |
| Geographic Progression | Southwest to Northeast | Regional timing |
| Southern US Peak | December-January | Earlier onset |
| Northeastern US Peak | April-May | Later onset |
| Post-Vaccine Pattern | Biennial cycling | Odd/even year variation |
| Odd Years | Higher incidence | Increased activity |
| Even Years | Lower incidence | Decreased activity |
| Current Trend (2026) | Even year – lower activity | Reduced transmission |
| Climate Factors | Temperature/humidity effects | Environmental influence |
Data Source: CDC NREVSS National and Regional Surveillance
In the United States, annual community outbreaks of rotavirus infections typically occurred during late fall, winter, and early spring in the pre-vaccine era, with marked geographic progression from southwest to northeast regions. Southern states experienced peak activity during December and January, while northeastern states saw highest incidence in April and May, creating a characteristic wave pattern across the continental United States lasting approximately five months annually.
Post-vaccine epidemiology transformed these patterns dramatically, with emergence of a unique biennial cycle where odd-numbered years (2009, 2011, 2013, 2021, 2023, 2025) showed elevated rotavirus incidence compared to even-numbered years (2008, 2010, 2012, 2022, 2024, 2026). This biennial pattern reflects annual fluctuations in susceptible children and transmission efficiency, with vaccination rates below 80% allowing accumulation of susceptible birth cohorts over two-year periods. Climate factors including temperature and humidity continue influencing rotavirus survival in the environment and transmission dynamics, though vaccination has substantially dampened the amplitude of seasonal peaks observed in pre-vaccine years.
Rotavirus Vaccine Safety in the US 2026
| Safety Parameter | Risk Assessment | Monitoring Data |
|---|---|---|
| Overall Safety Profile | Excellent safety | Extensive monitoring |
| Common Mild Reactions | Irritability, diarrhea | Self-limited |
| Intussusception Risk | 1-6 cases per 100,000 | Modestly elevated |
| Intussusception Timing | 7-10 days post-dose | Peak risk period |
| Age 8-11 Weeks Risk | First dose period | Increased vigilance |
| RotaTeq Intussusception Rate | 1 per 65,287 dose-1 | Very low absolute risk |
| Rotarix Intussusception Rate | 5.3 per 100,000 | Two-dose series |
| Benefit-Risk Ratio | Strongly favorable | Benefits outweigh risks |
| Contraindications | SCID, previous intussusception | Specific conditions |
Data Source: Vaccine Safety Datalink, CDC Vaccine Safety Surveillance
A modestly elevated risk of intussusception (approximately 1-6 cases per 100,000 vaccine recipients) has been noted in some international settings and in the United States, representing the primary safety concern with rotavirus vaccination. Intussusception involves bowel telescoping where one intestinal segment slides into an adjacent segment, creating potential obstruction requiring medical or surgical intervention. Post-licensure surveillance identified increased intussusception rates among infants 8-11 weeks old when first doses are typically administered.
Vaccine Safety Datalink analyses quantified attributable risk at 5.3 per 100,000 infants vaccinated with two doses of Rotarix and an upper limit of one case per 65,287 RotaTeq dose-1 recipients, representing extremely low absolute risk. Given the magnitude of declines in rotavirus disease compared with this small increase in intussusception, the benefits of rotavirus vaccination are considered to outweigh the increased risk. Contraindications include severe combined immunodeficiency (SCID), history of intussusception, and severe allergic reactions to previous doses, with infants allergic to latex advised against receiving Rotarix due to latex in the applicator.
Rotavirus Healthcare Costs in the US 2026
| Cost Category | Pre-Vaccine Era | Post-Vaccine Era |
|---|---|---|
| Annual Total Burden | $1 billion | Reduced substantially |
| Median Hospitalization Cost | $3,581 per child | Fewer hospitalizations |
| Annual Hospitalization Costs | $91 million | Dramatically reduced |
| Median ED Visit Cost | $574 per child | Fewer ED visits |
| Annual ED Costs | $192 million | Substantially decreased |
| Annual Healthcare Savings | Not applicable | $121-231 million |
| Prevented Hospitalization Value | Not applicable | 40,000-50,000 annually |
| Societal Cost Savings | Not applicable | $2.7 trillion cumulative |
| Cost Per Prevented Case | Not applicable | Highly cost-effective |
Data Source: CDC Economic Analysis, MarketScan Database, State Inpatient Database
Economic analyses of rotavirus disease demonstrate extraordinary cost savings achieved through vaccination programs. In the pre-vaccine era, annual costs totaled approximately $1 billion when accounting for direct medical expenses and indirect costs including parental work absences and long-term complications. The median hospitalization cost of $3,581 per child generated $91 million in annual hospitalization expenses based on 2009 dollars, while emergency department visits at $574 per child added $192 million annually.
Post-vaccine economic impact includes annual healthcare cost reductions of $121-231 million through prevented hospitalizations, emergency visits, and outpatient care. The 40,000-50,000 hospitalizations prevented annually translate to direct savings exceeding $140 million in hospitalization costs alone, excluding emergency department savings and indirect societal benefits. Cumulative societal cost savings from childhood vaccination programs including rotavirus are estimated at $2.7 trillion, demonstrating the profound economic value of immunization beyond individual health benefits. Cost-effectiveness analyses consistently show rotavirus vaccination provides excellent value, with benefits far exceeding vaccine procurement and administration costs.
Rotavirus Disparities in the US 2026
| Disparity Category | Coverage Rate | Impact |
|---|---|---|
| Overall National Average | 71.8% | Baseline coverage |
| American Indian/Alaska Native | 61.5% | Lowest coverage |
| Hispanic Children | Variable by region | Access challenges |
| Black Children | Lower than whites | Health equity gap |
| Uninsured Children | Significantly lower | Financial barriers |
| Medicaid-Insured Children | Lower than private | System challenges |
| Metropolitan Areas | Higher coverage | Better access |
| Non-Metropolitan Areas | 1-2% lower | Rural disparities |
| Foreign-Born Children | Lower completion | Immigration barriers |
Data Source: CDC National Immunization Survey, Racial and Ethnic Health Disparities
Racial and ethnic disparities are particularly evident among American Indian and Alaska Native children with 61.5% receiving rotavirus vaccination, representing a 10 percentage point gap below the national average and significantly lower than white children’s coverage rates. Black and Hispanic children also experience lower vaccination rates compared to non-Hispanic white children, reflecting systemic healthcare access barriers, insurance coverage gaps, and geographic distribution of vulnerable populations in underserved areas.
Insurance status creates substantial coverage disparities, with uninsured children and Medicaid-insured children showing significantly lower completion rates compared to privately insured children. The Vaccines for Children (VFC) program provides no-cost vaccines to eligible children, yet enrollment challenges and awareness gaps prevent universal access for all qualifying families. Geographic disparities show metropolitan areas achieving higher vaccination coverage than non-metropolitan and rural areas, where healthcare provider shortages and travel distances create additional barriers. Foreign-born children face unique challenges including delayed healthcare access, language barriers, and unfamiliarity with the American vaccination schedule.
Rotavirus Genotype Distribution in the US 2026
| Genotype | Pre-Vaccine Era | Post-Vaccine Era |
|---|---|---|
| G1P[8] | Predominant strain | Reduced prevalence |
| G2P[4] | Common strain | Variable circulation |
| G3P[8] | Common strain | Continued detection |
| G4P[8] | Common strain | Ongoing presence |
| G9P[8] | Emerging strain | Established circulation |
| G12P[8] | Rare pre-vaccine | Now predominant |
| Novel Reassortants | Occasional detection | Ongoing monitoring |
| Vaccine Coverage | Not applicable | Effective against variants |
| Strain Surveillance | Essential monitoring | Continued importance |
Data Source: CDC National Rotavirus Strain Surveillance System, NVSN Genotyping
In the pre-vaccine era, five distinct G and P combinations were most prevalent: G1[P8], G2[P4], G3P[8], G4P[8], and G9P[8], with G1P[8] serving as the dominant strain causing the majority of severe disease. Vaccine formulations specifically targeted these common circulating strains, with RotaTeq containing five reassortant viruses expressing G1, G2, G3, G4, and P[8] antigens, while Rotarix uses an attenuated G1P[8] strain capable of providing cross-protection against multiple genotypes.
Post-vaccine genotype surveillance revealed G12P[8] becoming a predominant strain among severe rotavirus gastroenteritis cases in recent years, though vaccine effectiveness against this non-vaccine genotype remains high due to cross-protective immunity. Secular variation in strain distribution occurs geographically, with different regions experiencing varying predominance of specific genotypes during individual seasons. Strain monitoring through the National Rotavirus Strain Surveillance System continues identifying novel reassortants and unusual genotypes, ensuring vaccine formulations maintain effectiveness against evolving viral populations. Reassortment events involving human and animal rotavirus genes occur occasionally, requiring ongoing surveillance to detect potential vaccine-escape variants.
Rotavirus Diagnosis in the US 2026
| Diagnostic Method | Sensitivity | Application |
|---|---|---|
| Enzyme Immunoassay (EIA) | 90-100% | Most widely used |
| RT-PCR | High sensitivity | Research/surveillance |
| Quantitative RT-PCR | Very high | Viral load measurement |
| Multipathogen Panels | High throughput | Comprehensive testing |
| Latex Agglutination | Lower sensitivity | Rapid bedside test |
| Electron Microscopy | High specificity | Research settings |
| Genotyping | Strain identification | Surveillance only |
| Clinical Diagnosis | Not specific | Presumptive diagnosis |
| Stool Specimen | Preferred sample | High viral concentration |
Data Source: CDC Laboratory Testing Guidelines, Diagnostic Methods
Enzyme immunoassay (EIA) represents the most widely available method for rotavirus antigen detection in stool, with several commercial kits available that are inexpensive, easy to use, rapid, and highly sensitive (approximately 90-100%), making them suitable for both clinical diagnosis and surveillance applications. EIA tests detect antigens common to all group A rotaviruses, providing reliable diagnosis without requiring strain-specific identification. Turnaround times of several hours allow same-day results influencing clinical management decisions.
Molecular methods including reverse transcription polymerase chain reaction (RT-PCR) and quantitative RT-PCR offer higher sensitivity and viral load quantification, primarily used in research settings and public health surveillance rather than routine clinical practice. Multipathogen diagnostic panels like the Luminex Gastrointestinal Pathogen Panel and FilmArray GI Panel enable simultaneous detection of multiple enteric pathogens including rotavirus, norovirus, bacterial pathogens, and parasites in a single test. Latex agglutination tests provide rapid results but demonstrate lower sensitivity and specificity compared to EIA methods. Genotyping and sequencing enable strain characterization critical for surveillance purposes but remain unnecessary for clinical management.
Rotavirus Treatment in the US 2026
| Treatment Category | Intervention | Effectiveness |
|---|---|---|
| Primary Treatment | Oral rehydration therapy | Highly effective |
| Severe Dehydration | Intravenous fluids | Life-saving |
| Hospitalization | Supportive care | Monitoring/IV therapy |
| Zinc Supplementation | Recommended in some countries | Reduces duration |
| Antibiotics | Not indicated | Ineffective for virus |
| Antiviral Medications | None approved | Not available |
| Ondansetron | Anti-vomiting medication | Facilitates oral intake |
| Probiotics | Adjunctive therapy | Variable evidence |
| Breastfeeding | Continued during illness | Maintains hydration |
Data Source: CDC Clinical Management Guidelines
Treatment is nonspecific and consists primarily of oral rehydration therapy to prevent dehydration. For people with healthy immune systems, rotavirus disease is self-limited, lasting only a few days. Oral rehydration solutions containing appropriate electrolyte concentrations represent the cornerstone of management, with frequent small volume administration overcoming vomiting that might otherwise prevent adequate fluid intake. World Health Organization and CDC-recommended solutions provide optimal sodium, potassium, and glucose ratios for intestinal absorption.
Severe dehydration requires intravenous fluid therapy administered in emergency departments or hospital settings, with electrolyte monitoring ensuring appropriate sodium, potassium, and acid-base balance. Hospitalization rates have declined dramatically post-vaccination, though children presenting with severe dehydration, shock, or electrolyte imbalances require intensive supportive care. Zinc supplementation shows benefits in developing countries by reducing diarrhea duration, though routine use in the United States remains uncommon. Antibiotics provide no benefit for viral gastroenteritis and should be avoided except when bacterial co-infection is suspected. Ondansetron as an anti-emetic medication helps reduce vomiting, facilitating oral rehydration success and potentially preventing hospitalization.
Disclaimer: The data research report we present here is based on information found from various sources. We are not liable for any financial loss, errors, or damages of any kind that may result from the use of the information herein. We acknowledge that though we try to report accurately, we cannot verify the absolute facts of everything that has been represented.