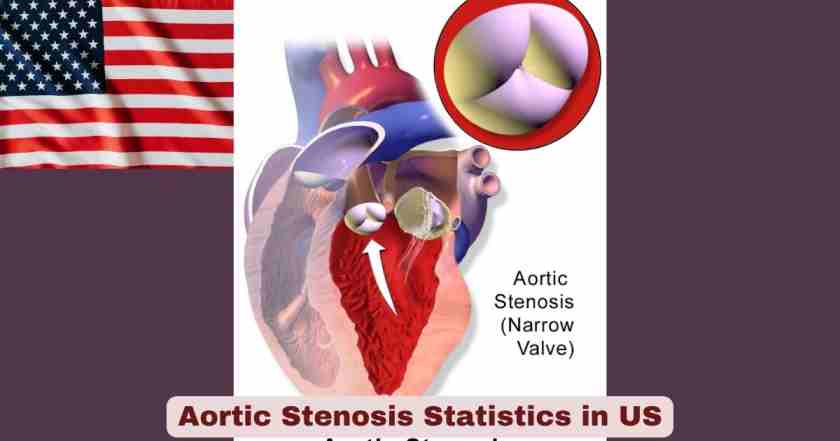
What Is Aortic Stenosis?
Aortic stenosis (AS) is the most common valvular heart disease in the developed world and one of the most consequential cardiovascular conditions affecting older Americans. It occurs when the aortic valve — the one-way gate between the heart’s left ventricle and the aorta — becomes progressively narrowed, forcing the heart to work significantly harder to pump blood through the restricted opening. Over time, this pressure overload causes the heart muscle to thicken, weaken, and ultimately fail. The disease is predominantly calcific and degenerative in nature in high-income countries, meaning it develops slowly over decades as calcium deposits accumulate on the valve leaflets, stiffening them until they can no longer open fully. In the United States, aortic stenosis is the second most common valvular lesion, affecting approximately 5% of the population at age 65 and rising sharply with advancing age — from 0.2% in the 50–59 age group to 9.8% in those aged 80–89 years. The landmark PREVUE-VALVE study, presented at TCT® 2025 in October 2025, found that at least 4.7 million Americans aged 65–85 currently live with moderate or greater valvular heart disease, and that aortic stenosis (at 3.1% prevalence in that age group) is the second most common form — yet most affected patients remain unaware they have the condition, making underdiagnosis and delayed treatment one of the most serious ongoing challenges in US cardiovascular medicine.
What distinguishes aortic stenosis from many other serious heart conditions is the deceptive nature of its progression. Patients can remain completely asymptomatic for years while the disease advances silently, and the heart compensates through a process of hypertrophy and remodeling that eventually exhausts itself. The critical inflection point is the onset of symptoms — classically chest pain (angina), fainting (syncope), or shortness of breath (dyspnea on exertion), often collectively called the “classic triad of AS.” Once symptoms appear, the prognosis without treatment deteriorates rapidly: without aortic valve replacement, as many as 50% of symptomatic severe AS patients do not survive beyond 2 years. This grim natural history makes AS one of the most urgent indications for intervention in all of cardiology. The treatment landscape, however, has been transformed over the past decade by the advent of Transcatheter Aortic Valve Replacement (TAVR) — a catheter-based procedure that implants a new valve without open-heart surgery, dramatically expanding the eligible patient population and reshaping how aortic stenosis is managed from diagnosis through intervention. The landmark EARLY TAVR trial, published in the New England Journal of Medicine in October 2024, now provides the strongest evidence yet that early intervention in even asymptomatic severe AS cuts the composite risk of death, stroke, or hospitalization in half.
Interesting Facts: Aortic Stenosis in the US
| Fact | Detail |
|---|---|
| Common name | Aortic stenosis (AS) — also called aortic valve stenosis or calcific aortic valve disease |
| Most common cause in high-income countries | Calcific / degenerative aortic stenosis — calcium deposits on valve leaflets |
| 2nd most common cause (congenital) | Bicuspid aortic valve (BAV) — affects 1–2% of general population; 31.9% of surgical aortic valve cases |
| Ranking among US valvular diseases | 2nd most common valvular lesion in the United States |
| US AS prevalence at age 65 | ~5% of the population at age 65 |
| US AS prevalence at age 80–89 | ~9.8% of that age group |
| US AS prevalence at age 70–79 | ~3.9% |
| US AS prevalence at age 60–69 | ~1.3% |
| US AS prevalence at age 50–59 | ~0.2% |
| Severe AS prevalence in 75+ population | 3.4% in Europe and USA combined |
| PREVUE-VALVE study (TCT 2025) | At least 4.7 million Americans aged 65–85 live with moderate or greater valvular heart disease |
| Aortic stenosis in PREVUE-VALVE | 3.1% prevalence — second most common VHD in 65–85 age group |
| AS patients unaware of condition | Majority — most patients with VHD are unaware of their condition |
| US total AS patients (older estimate) | Up to 1.5 million Americans suffer from AS; ~500,000 have severe AS; ~250,000 are symptomatic with severe AS |
| Severe AS incidence (community-based) | 52.5 per 100,000 patient-years (stable over 20 years; Mayo Clinic/Olmsted County) |
| Age-standardized AS prevalence trend | Increased from 589 to 754 per 100,000 during 2013–2019 (integrated health system study) |
| AS projected doubling by 2050 | Global AS prevalence expected to double by 2050 due to aging populations |
| Symptomatic severe AS — untreated prognosis | ~50% do not survive beyond 2 years without valve replacement |
| Average survival after severe symptomatic AS onset — untreated | 2 to 3 years on average |
| AVR undertreatment rate | >40% undertreatment remains prevalent even in modern era (Mayo Clinic study) |
| Mortality trend (US 1999–2020) | 267,515 AS deaths among Americans >65 from 1999–2020; AAMR declined from 28.00 to 23.69 per 100,000 |
| Deaths: AS + hypertension (1999–2020) | 99,250 deaths from concurrent AS and hypertension in US older adults |
| TAVR dominance | TAVR has become the predominant intervention for severe symptomatic AS in the US regardless of surgical risk |
| TAVR first FDA approval | November 2011 (inoperable patients); 2012 (high-risk); 2019 (low-risk patients) |
| EARLY TAVR trial result (NEJM, October 2024) | Early TAVR in asymptomatic severe AS: hazard ratio 0.50 — cut composite risk of death/stroke/hospitalization in half |
| TAVR vs SAVR at 5 years (Evolut Low Risk) | Comparable outcomes: 15.5% vs 16.4% all-cause mortality/disabling stroke — statistically non-inferior |
Source: ESC Journal of Cardiology Practice (epidemiology review), PMC Age Differences in AS (January 2025), MDPI Trends in AS Mortality (November 2025), CRF PREVUE-VALVE study (October 27, 2025 — TCT 2025), ACC/European Heart Journal (Benfari et al. 2024), John Muir Health facts and figures, NEJM EARLY TAVR (October 2024), JACC Evolut Low Risk 5-year (2025), JTCVS Structural (2024), Surgeries MDPI (December 2024)
The most striking number in this table is one that rarely gets the attention it deserves: >40% undertreatment. In a 20-year community study of severe AS at the Mayo Clinic, more than four in ten patients who met the clinical criteria for aortic valve replacement did not receive it — despite growing procedure volumes and despite severe AS’s well-established, life-threatening natural history. This is not primarily a problem of treatment availability or technology; it reflects the diagnostic gap created by asymptomatic disease progression, patient hesitancy, access disparities, and the complexity of risk-benefit communication for older, frailer patients. The PREVUE-VALVE finding that most VHD patients — including those with AS — are unaware of their condition underscores the same problem from a population health perspective: the disease is progressing silently in millions of Americans who have not been screened or diagnosed.
The mortality trend data from 1999 to 2020 tells a more encouraging story: over two decades of data tracking 267,515 AS-related deaths among Americans over 65, the age-adjusted mortality rate declined from 28.00 to 23.69 per 100,000 — a meaningful improvement that likely reflects both better medical management and the progressive rollout of TAVR from 2011 onward. But that improvement is set against the backdrop of an aging US population that will drive absolute case numbers significantly higher even as age-adjusted rates improve. The projected doubling of global AS prevalence by 2050 is not a distant abstraction — it is an actionable planning reality for US cardiovascular medicine, public health infrastructure, and healthcare financing.
Aortic Stenosis Symptoms & Clinical Profile
| Symptom / Clinical Feature | Detail |
|---|---|
| Classic triad of AS symptoms | Angina (chest pain), Syncope (fainting), Dyspnea (shortness of breath on exertion) |
| Asymptomatic phase | Disease can progress silently for years to decades before symptoms develop |
| Typical symptom onset age | 65–75 years for calcific degenerative AS |
| Symptom onset — bicuspid valve | Often earlier — 40s–60s — due to congenital valve abnormality |
| Angina / chest pain | Occurs with exertion when narrowed valve limits cardiac output increase |
| Syncope / near-fainting | Especially on exertion; results from inability to increase cardiac output |
| Exertional dyspnea | Shortness of breath on activity — often first and most common symptom |
| Fatigue | Due to reduced cardiac output; often mistaken for normal aging |
| Heart failure symptoms | Fluid retention, breathlessness at rest — indicate advanced disease |
| Heart murmur | Systolic ejection murmur heard on auscultation — primary clinical detection method |
| Reduced exercise tolerance | Increasing limitation as disease progresses |
| Asymptomatic severe AS prognosis | 25% of asymptomatic patients required AVR or died within 1 year; 75% within 5 years in observational study |
| Symptomatic severe AS — untreated 2-year survival | ~50% die within 2 years of symptom onset without intervention |
| Cognitive impairment (emerging) | Growing body of evidence linking severe AS to cognitive decline and dementia risk |
| Concurrent cardiac amyloidosis | Up to 15% of severe AS patients have coexisting cardiac amyloidosis — significantly worsens prognosis (2.6× higher mortality) |
| Echocardiogram — primary diagnostic tool | Transthoracic echocardiogram (TTE) — the primary imaging test to diagnose and grade AS severity |
| Echocardiographic grading | Mild, moderate, severe — based on valve area, mean gradient, peak velocity |
| Severe AS diagnostic criteria | Aortic valve area ≤1 cm², aortic valve area index ≤0.6 cm²/m², mean gradient ≥40 mmHg, peak velocity ≥4 m/s |
| Very low-gradient severe AS challenge | “Paradoxical low-flow, low-gradient AS” — difficult to diagnose; often associated with reduced LVEF |
Source: CRF PREVUE-VALVE report (October 27, 2025), John Muir Health facts and figures, NEJM EARLY TAVR (October 2024), JAHA meta-analysis on early AVR (2025), PubMed Ahmad et al. AS + cardiac amyloidosis (2025), European Heart Journal Benfari et al. (2024), JTCVS Structural (2024), PMC Age Differences in AS (January 2025)
Aortic stenosis’s clinical trajectory has a cruel internal logic. The heart compensates for the narrowing valve through hypertrophy — the muscle thickens to generate more pressure — and this compensation can be so effective that patients feel essentially normal for years while the valve area progressively shrinks and the pressure gradient across it grows. The classic symptom triad of angina, syncope, and dyspnea does not typically appear until the valve area has narrowed to below 1 cm² (from a normal area of 3–4 cm²) and the heart’s compensatory capacity is nearly exhausted. This means that by the time most patients notice something is wrong, the disease has been present and progressing for years. The EARLY TAVR trial’s finding that even asymptomatic severe AS patients randomized to clinical surveillance deteriorated rapidly — with 87% ultimately requiring valve replacement within a median of 3.8 years — quantifies just how compressed the natural history becomes once severe AS is established.
The 15% prevalence of cardiac amyloidosis among severe AS patients adds an important clinical complexity layer. Cardiac amyloidosis — in which amyloid protein deposits stiffen the heart muscle — is increasingly recognized as a co-pathology in elderly AS patients, and the 2025 meta-analysis by Ahmad et al. confirmed that patients with both conditions face 2.6× higher all-cause mortality compared to AS alone. This means that the growing clinical priority of routine amyloid screening alongside AS workup is not merely academic — it directly affects treatment planning and prognosis. The diagnosis of AS itself remains strikingly dependent on detecting the characteristic systolic ejection murmur on physical examination and then confirming severity by echocardiogram. The PREVUE-VALVE study’s finding that most affected patients are unaware of their condition suggests that community-based echo screening may be underutilized, and that the diagnostic gap is widest in patients who have not yet developed the overt symptoms that drive them to cardiology referral.
Aortic Stenosis Causes & Risk Factors in the US | Etiology Data
| Cause / Risk Factor | Data |
|---|---|
| Most common cause (US/developed world) | Calcific/degenerative aortic stenosis — calcium accumulation on valve leaflets over decades |
| Bicuspid aortic valve (BAV) — congenital | 31.9% of surgical aortic valve cases had BAV; general population prevalence 1–2% |
| Unicuspid aortic valve (UAV) | Present in 3.0% of surgical excision cases |
| Post-rheumatic AS | 11% of surgically excised valves had rheumatic disease — declining in developed countries |
| Age — primary risk factor | Prevalence increases exponentially with age: 0.2% (50–59) → 9.8% (80–89) |
| Sex — calcific AS | Slightly higher incidence in men (stable); incidence declined slightly in women over 20 years |
| Hypertension | Strongly associated; concurrent AS + hypertension caused 99,250 deaths 1999–2020 |
| Hyperlipidemia / high LDL | Associated with calcification process — though statin trials have not consistently slowed AS progression |
| Diabetes | Associated with increased AS risk and worse outcomes |
| Obesity | Associated with increased cardiovascular risk and AS |
| Smoking | Associated with increased cardiovascular risk and calcific AS development |
| Chronic kidney disease (CKD) | Associated with accelerated vascular and valve calcification |
| Race — non-Hispanic White | Higher prevalence of AS; Black patients have ~20% lower age-adjusted prevalence of aortic valve disease vs White patients |
| Geographic variation in US | Maine, Vermont, Iowa, New Hampshire, Oregon have higher AS mortality — older, whiter populations; also Midwest and South |
| Genetic factors — BAV | BAV has a polygenic basis with variants in NOTCH1, MYH6, GATA4, GATA5, GATA6, and others; hereditary within families |
| BAV associated syndromes | Marfan syndrome, Turner syndrome, Loeys-Dietz syndrome |
| Radiation therapy history | Prior chest radiation (e.g., for lymphoma/breast cancer) associated with accelerated valve calcification |
| Lifestyle exposure | Harsh winter climates, rural healthcare access barriers also associated with higher AS mortality in some US regions |
Source: PMC Age Differences in AS (January 2025), ESC Epidemiology of AS Review, ACC/European Heart Journal Benfari et al. (2024), PMC Concurrent AS and Hypertension (2025), MDPI Trends in AS Mortality (November 2025), CRF PREVUE-VALVE (October 27, 2025)
The etiology of aortic stenosis in the United States has undergone a structural shift over the past half century. Before modern cardiology’s understanding of calcific valve disease, rheumatic heart disease — caused by untreated streptococcal infection leading to immune-mediated valve damage — was a leading cause of AS. In contemporary US clinical data, only 11% of surgically excised valves show evidence of rheumatic disease, down dramatically from earlier decades, reflecting improvements in antibiotic treatment of streptococcal infections. What has replaced it as the overwhelmingly dominant pathway is calcific/degenerative AS: a slow, decades-long accumulation of calcium on the valve leaflets driven by the same mechanistic forces that produce atherosclerosis — lipid infiltration, oxidative stress, chronic inflammation, and mechanical stress at areas of high shear force on the valve. This is why the traditional cardiovascular risk factors (hypertension, hyperlipidemia, diabetes, smoking, obesity) are associated with AS risk: they accelerate the calcification process, even though the interventions that reduce cardiovascular events from atherosclerosis (statins, blood pressure medications) have disappointingly not been shown to consistently slow AS progression once it has begun.
The racial disparity data from PREVUE-VALVE deserves particular attention. The finding that Black Americans have approximately 20% lower age-adjusted prevalence of aortic valve disease compared to White Americans — driven largely by lower rates of calcific AS rather than differences in other valve diseases — is a real epidemiological signal, not a testing artifact. It may reflect differences in the inflammatory and metabolic pathways that drive calcification, differences in exposure to post-rheumatic disease (which is more common among Black patients and confers somewhat different AS risk), or both. Importantly, this lower prevalence does not mean Black patients face lower risk of poor outcomes when they do develop AS — access disparities, later-stage diagnosis, and underrepresentation in TAVR recipient populations mean Black patients with AS may actually receive less timely intervention, a health equity problem that CRF’s director David Cohen explicitly highlighted at TCT 2025.
Aortic Stenosis Treatment in the US | TAVR vs SAVR & Current Options
| Treatment | Description / Status | Key Outcomes Data |
|---|---|---|
| Surgical Aortic Valve Replacement (SAVR) | Open-heart surgery; removes and replaces native aortic valve — mechanical or bioprosthetic valve | 5-year survival ~80%; gold standard for low-risk younger patients |
| Transcatheter Aortic Valve Replacement (TAVR) | Catheter-based; valve delivered via femoral artery without open chest — now the predominant procedure in the US | Non-inferior to SAVR at 2 and 5 years across risk groups |
| TAVR FDA approval history | 2011 (inoperable), 2012 (high-risk), 2019 (expanded to low-risk patients) | Expanded eligibility revolutionized the field |
| TAVR vs SAVR — 5-year primary endpoint (Evolut Low Risk) | All-cause mortality/disabling stroke: TAVR 15.5% vs SAVR 16.4% (P = 0.69) — non-inferior | Valve durability and performance excellent in both arms |
| TAVR cardiovascular mortality at 5 years | 7.2% (TAVR) vs 9.3% (SAVR) — not statistically significant | Numerically favorable for TAVR; not statistically different |
| TAVR valve reintervention at 5 years | 3.3% (TAVR) vs 2.5% (SAVR) — P = 0.44 — not significantly different | Both groups: excellent valve durability at midterm |
| NOTION trial at 10 years | No significant differences in mortality, stroke, or MI between SAVR and TAVR | Structural valve deterioration lower after TAVR (13.9% vs 28.3%) |
| EARLY TAVR trial (NEJM, October 2024) | 901 patients randomized: Early TAVR vs clinical surveillance for asymptomatic severe AS | Primary endpoint: 26.8% TAVR vs 45.3% surveillance — HR 0.50 — superior |
| EARLY TAVR — death | 8.4% TAVR vs 9.2% surveillance — numerical benefit, not statistically significant for death alone | Death reduction trend favors early TAVR |
| EARLY TAVR — stroke | 4.2% TAVR vs 6.7% surveillance — meaningful reduction | Stroke risk reduced with early intervention |
| EARLY TAVR — unplanned CV hospitalization | 20.9% TAVR vs 41.7% surveillance — dramatic reduction | Primary driver of composite endpoint benefit |
| Surveillance group in EARLY TAVR | 87% of clinical surveillance patients ultimately required AVR within median 3.8 years | Confirms that “watchful waiting” leads to nearly universal eventual intervention |
| Age guideline — ACC | TAVR preferred for patients >65 years; SAVR for younger or anatomically unsuitable | Age is a key but not sole determinant |
| SAVR advantages | Preferred for: younger age, lower surgical risk, bicuspid anatomy, concomitant cardiac conditions needing repair | NOTION-2: SAVR superior for bicuspid patients (14.3% vs 3.9% events) |
| TAVR advantages | Less invasive, shorter recovery, suitable for high/intermediate-risk, transfemoral access | Vascular complications and arrhythmia (new pacemaker) more common with TAVR |
| Balloon aortic valvuloplasty (BAV) | Palliative option — not a replacement for AVR; used as bridge to TAVR | Short-term improvement only; high restenosis rate |
| Medical management only | For patients unwilling or unable to undergo AVR; no drug proven to slow or reverse AS | Diuretics, antihypertensives for symptom management only |
| AS with cardiac amyloidosis (AS-CA) | TAVR preferred over SAVR for AS-CA patients — 50% lower mortality vs medical therapy | TAVR associated with lower all-cause mortality than both medical therapy (RR 0.50) and SAVR (RR 0.41) |
| Quality of life after AVR | Mean KCCQ score of 88.3 (TAVR) and 88.5 (SAVR) at 5 years — sustained, comparable improvement | Both procedures deliver excellent QoL outcomes at midterm |
Source: JACC (Evolut Low Risk 5-year outcomes, 2025), NEJM (EARLY TAVR, October 28, 2024), Annals of Thoracic Surgery (2024), JTCVS Structural and Endovascular (2024), Surgeries MDPI (December 2024), PubMed Ahmad et al. AS + cardiac amyloidosis (April 2025), Cardiovasculardiseasehub.com
The treatment of aortic stenosis has undergone the most dramatic transformation of any cardiac condition over the past 15 years. Before TAVR, elderly or frail patients who were deemed too high-risk for open-heart surgery essentially had no good options — they were managed medically with symptomatic relief, with the knowledge that median survival after symptom onset was measured in months to a couple of years. The first FDA approval of TAVR in November 2011 changed that calculus completely. Within a decade, TAVR had expanded from inoperable patients to high-risk, intermediate-risk, and finally low-risk patients (2019), and had become the predominant intervention for severe symptomatic AS in the United States, regardless of surgical risk. The 5-year data from the Evolut Low Risk trial — confirming non-inferiority of TAVR versus SAVR with comparable valve durability — cemented TAVR’s clinical legitimacy even for patients who would previously have been considered ideal open-surgery candidates.
The EARLY TAVR trial’s October 2024 publication in the NEJM may be the most consequential piece of AS clinical evidence in a generation. By demonstrating that early TAVR in asymptomatic severe AS patients cuts the composite risk of death, stroke, or unplanned hospitalization in half (hazard ratio 0.50), it challenges the previous standard of “watchful waiting” for asymptomatic patients and opens the possibility of a paradigm shift toward earlier intervention. The finding that 87% of the clinical surveillance group eventually underwent AVR anyway — just later, and after more events — makes the cost-benefit case for early treatment particularly compelling. Importantly, the stroke reduction from 6.7% to 4.2% in the early TAVR group is clinically significant in its own right, because stroke in this elderly population has devastating consequences. The EARLY TAVR results will likely drive guideline revisions and affect how cardiologists counsel asymptomatic AS patients for years to come.
Aortic Stenosis Prevention & Outlook in the US
| Prevention / Outlook Area | Current Status |
|---|---|
| No proven prevention for calcific AS | No drug or intervention has been shown to prevent or reverse calcific valve disease |
| Statins and AS | Despite atherosclerosis-AS overlap, randomized trials have not shown statins slow AS progression |
| Blood pressure control | Hypertension management recommended — reduces cardiovascular comorbidity; does not reverse valve disease |
| Cardiovascular risk factor reduction | Standard measures (smoking cessation, diabetes control, weight management, exercise) reduce overall CV risk |
| Prevention of rheumatic AS | Prompt antibiotic treatment of streptococcal pharyngitis (strep throat) — effective prevention of rheumatic fever |
| Echocardiographic screening | No universal screening recommendation in US currently; PREVUE-VALVE advocates for broader screening access |
| AS mortality trend — positive | AAMR declined from 28.00 to 23.69 per 100,000 in >65 population from 1999–2020 |
| TAVR procedure growth | AVR volumes grew significantly over 20-year study period; time to treatment decreased from 1.3 years (1997–2000) to 0.5 years (2013–2016) |
| Undertreatment still >40% | Despite AVR growth, >40% of severe AS patients still do not receive valve replacement |
| Health equity gap | Black patients have ~20% lower AS prevalence but face access barriers to timely intervention |
| Regional mortality disparities | Southern and Midwestern US states have higher uninsured rates, obesity, hypertension — contributing to AS burden |
| TAVR in lower-risk, younger patients | Active research and guideline evolution; durability data beyond 10 years still limited |
| AS prevalence set to double by 2050 | Aging global population; patients 70+ will double or triple in developed countries |
| TAVR cost-effectiveness | Favorable cost-effectiveness established for high-risk; growing evidence for intermediate and low-risk |
| AS + cognitive decline research | Emerging evidence of bidirectional relationship; cerebral microemboli during TAVR are studied |
| VHD screening initiative | TCT 2025 discussion on structured VHD screening programs for 65+ population |
| Valve-in-valve (ViV) TAVR | Growing option for bioprosthetic valve failure — SAVR patients can receive TAVR-in-SAVR for degenerated surgical valves |
| Bicuspid AS treatment evolution | NOTION-2 showed SAVR superior for bicuspid patients at 1 year; long-term data from TAVR in bicuspid patients accumulating |
Source: MDPI Trends in AS Mortality (November 2025), CRF PREVUE-VALVE (TCT 2025, October 27, 2025), ACC / European Heart Journal Benfari et al. (2024), PMC Age Differences in AS (January 2025), JTCVS Structural (2024), Annals of Thoracic Surgery (2024)
The prevention story for calcific aortic stenosis is one of medicine’s genuine frustrations. The mechanistic overlap between AS and atherosclerosis — both driven by lipid infiltration, inflammation, and calcification — led to a compelling hypothesis that statins, which dramatically reduce cardiovascular events from atherosclerosis, should also slow AS progression. Randomized trials including ASTRONOMER and SEAS unambiguously refuted that hypothesis: statins do not slow the hemodynamic progression of established calcific AS. This means that unlike coronary artery disease, where risk factor modification can genuinely prevent or regress disease, established calcific AS has no pharmacological brake — once the process is underway, the valve will continue to narrow until it requires replacement. The clinical implication is stark: early detection matters more than any preventive medication, because the window in which careful monitoring, timely surgery, and appropriate lifestyle management can most benefit patients is while the disease is still progressing toward — rather than through — the threshold for intervention.
The >40% undertreatment rate that persists in contemporary AS management is perhaps the most actionable finding in the entire body of recent AS research. It is not primarily a technology or efficacy problem — TAVR and SAVR both produce excellent outcomes, and procedural safety has improved dramatically. It is a recognition, referral, and access problem. Patients who don’t know they have AS can’t be treated. Patients who know they have AS but face geographic, financial, or health literacy barriers to a specialized valve center don’t get treated. Patients whose cardiologist practices watchful waiting conservatively in a pre-EARLY-TAVR world don’t get treated promptly. The PREVUE-VALVE study’s call for broader VHD screening programs, the EARLY TAVR data supporting earlier intervention, and the declining time-to-treatment trend (from 1.3 years in the late 1990s to 0.5 years in the 2010s) all point in the same direction: the most impactful changes in AS outcomes over the next decade will come less from new device innovation and more from systematic improvement in who gets diagnosed, when, and how quickly they reach effective treatment.
Disclaimer: The data research report we present here is based on information found from various sources. We are not liable for any financial loss, errors, or damages of any kind that may result from the use of the information herein. We acknowledge that though we try to report accurately, we cannot verify the absolute facts of everything that has been represented.